The Adrenaline Rush: COVID-19 & The Vaccine
By William Carroll, System VP of Pharmacy Services, Rochester Regional Health
Are you an adrenaline junky? Do you like rollercoasters? The slow tick as you climb higher and higher and then the sudden drop at warp speed truly takes your breath away. Reaching the bottom only for a split second to catch your breath before suddenly climbing again. What a rush!!! What about bungee jumping….feeling the free fall, weightlessness until the harness and cord abruptly awaken you out of your peaceful trance. Or maybe the thought of walking onto a stage and sitting down at a nearly 9 foot Steinway Model D grand piano… 1000s of eyes awaiting your delicate touch of the first keys… taking a deep breath before you give perhaps the best performance of your life! Each scenario may evoke a similar response… increased heart rate, respiration, perspiration. Adrenaline overload!
Some of you may not be as excited as I am and may describe one or more of those scenarios as crazy, insane, ridiculous. A safe, controlled, predictable environment is maybe more your speed. Why would anyone willingly subject themselves to this type of sudden unexpected, uncontrollable chaos?
Well – living through not 1 but 2 COVID surges and being in the midst of a third may feel very much like a rollercoaster ride, bungee jump, or public performance for some of us. COVID has forced healthcare leaders, from the largest health systems down to the smallest independent hospital, to rethink our typical day to day operations. Not only have we been forced to accommodate a massive increase in hospital patient volumes but also ensure we keep our team members safe and resilient while awaiting the next turn on this ride.
The Rochester Regional Health (RRH) Vaccine Reconciliation Web Application provides a way for crucial state-required data points to be collected in a simple and accurate manner.
Hope
It almost seemed like a hopeless endeavor as our patient volumes vacillated up and down with each surge,until a glimmer of hope shone its face on a cold, snowy, upstate NY evening in December 2020. On Dec. 11, the U.S. Food and Drug Administration (FDA) authorized the first COVID-19 vaccine for use in the country. Finally, it seemed this might not be a never-ending rollercoaster after all. You could feel the renewed sense of hope in the organization as we prepared for vaccine arrival.
Vaccine manufacturers announced that they had tens of millions of doses ready to disperse, and we developed plans to ensure we would be prepared to administer them quickly and efficiently. We had spent the preceding weeks preparing for this moment based on information we had prior to the EUA approval – ultra-cold freezers on order, clinic operating models developed, per diem staff hired, toolkits prepared. We were re-invigorated by this historical moment and the incredible role we were elected to play to help end a worldwide pandemic. We were ready for the vaccine floodgates to open! However, we were not prepared for the many patient eligibility criteria and data request changes that would occur on a weekly basis over the coming weeks.
Evolve
There were several challenges associated with managing COVID-19 vaccine. Ultracold deep freezers were on backorder, along with syringes and needles. The need to maintain cold chain integrity posed its only challenges that we were indeed able to overcome with the development of clear standard operating procedures. However, one of the greatest challenges for many health systems was tracking their state-requested data points around received, administered, and wasted vaccines. In the initial stages, this was easier when there was only one manufacturer available, only first doses being administered, and very minimal patients eligible to receive the vaccine. However, once multiple vaccines received EUA approval status, second dose administration began, and eligibility criteria continued to expand, tracking vaccine data became a challenge in order to meet the state’s requests.
At one point, we were hosting Pfizer®, Moderna®, and Janssen®, first and second dose, employee and patient vaccinations at 10+ different hospital and community mass clinics simultaneously. How do you keep track of: Who has vaccine? How much do they have in inventory? Who was vaccinated at the clinic and was it a first or second dose? How many doses were wasted?
Teamwork
To find a solution, we leveraged the amazing talent in our organization. Key pharmacy and IT business intelligence leaders found a simple yet effective solution for our health system: a web-based application. The Rochester Regional Health (RRH) Vaccine Reconciliation Web Application (screenshot below) provides a way for crucial state-required data points to be collected in a simple and accurate manner. At the end of each clinic, the vaccine clinic leadership team completes this web application survey. The data is reviewed by a dedicated resource who compiles the information and reports into the state via their online tool.
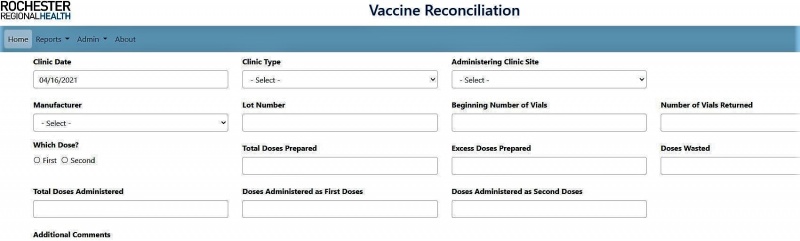
The creation of this web application has led to a much more efficient and accurate inventory management process for COVID-19 vaccine. We are able to update the application to capture new data requests and no longer maintain multiple Excel spreadsheets and paper logs to track inventory. Lastly, we have gone from having daily vaccine reconciliation calls to rare ad hoc calls to address changes in state data requirements.
Now, after 2 COVID-19 surges and four months experience of administering COVID-19 vaccine, it no longer feels like we are riding a mile high rollercoaster for the first time. We are taking each rise and fall in stride and learning to let out a squeal of delight when we accomplish something great! It’s our duty as healthcare leaders – to remain hopeful, continue to be flexible and evolve, and function as a team in order to meet our patient and community needs. I don’t expect this ride to end anytime soon, but we can definitely buckle up and make the most of it.



